8 results
Neural response to monetary and social rewards and familial risk for psychopathology in adolescent females
- Clare C. Beatty, Kelly Gair, Joy Anatala, Daniel N. Klein, Greg Hajcak, Brady D. Nelson
-
- Journal:
- Psychological Medicine , First View
- Published online by Cambridge University Press:
- 04 January 2024, pp. 1-11
-
- Article
- Export citation
-
Background
Adolescence is a key developmental period for the emergence of psychopathology. Reward-related brain activity increases across adolescence and has been identified as a potential neurobiological mechanism of risk for different forms of psychopathology. The reward positivity (RewP) is an event-related potential component that indexes reward system activation and has been associated with both concurrent and family history of psychopathology. However, it is unclear whether the RewP is also associated with higher-order psychopathology subfactors and whether this relationship is present across different types of reward.
MethodsIn a sample of 193 adolescent females and a biological parent, the present study examined the association between adolescent and parental psychopathology subfactors and adolescent RewP to monetary and social reward.
ResultsResults indicated that the adolescent and parental distress subfactors were negatively associated with the adolescent domain-general RewP. The adolescent and parental positive mood subfactors were negatively associated with the adolescent domain-general and domain-specific monetary RewP, respectively. Conversely, the adolescent and parental fear/obsessions subfactors were positively associated with the adolescent domain-general RewP. The associations between parental and adolescent psychopathology subfactors and the adolescent RewP were independent of each other.
ConclusionsThe RewP in adolescent females is associated with both concurrent and parental psychopathology symptoms, suggesting that it indexes both severity and risk for higher-order subfactors.
Integrated efficacy analysis from phase 3 studies of investigational microbiome therapeutic, SER-109, in recurrent Clostridioides difficile infection
- Matthew Sims, Michael Silverman, Thomas Louie, Elaine Wang, Colleen Kraft, Mayur Ramesh, Tatiana Bogdanovich, Kelly Brady, David Lombardi, Asli Memisoglu, Ananya De, Brooke Hasson, Christine Lee, Paul Feuerstadt, Darrell Pardi, Colleen Kelly, Peter Daley, Godson Oguchi, Barbara McGovern, Lisa Von Moltke
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, p. s5
-
- Article
-
- You have access Access
- Open access
- Export citation
-
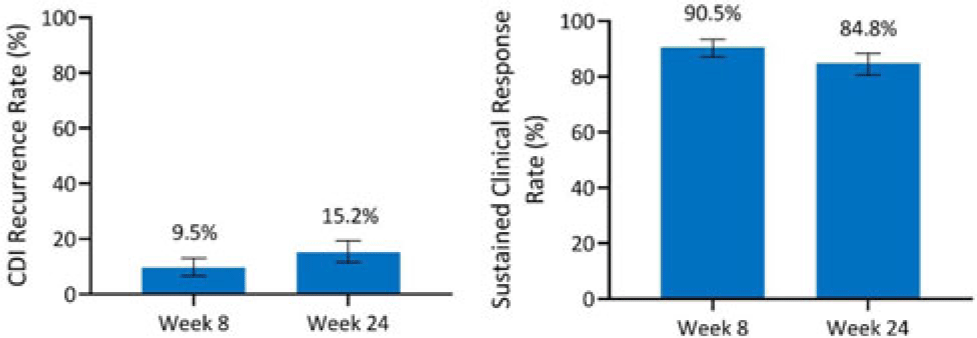
Background: Antibiotics alone are often insufficient to treat recurrent C. difficile infection (rCDI) because they have no activity against C. difficile spores that germinate within a disrupted microbiome. SER-109, an investigational, oral, microbiome therapeutic comprised of purified Firmicutes spores, was designed to reduce rCDI through microbiome repair. We report an integrated efficacy analysis through week 24 for SER-109 from phase 3 studies, ECOSPOR III and ECOSPOR IV. Methods: ECOSPOR III was a randomized, placebo-controlled phase 3 trial conducted at 56 US or Canadian sites that included 182 participants with ≥2 CDI recurrences, confirmed via toxin EIA testing. Participants were stratified by age (<65 years or ≥65 years) and antibiotic regimen (vancomycin, fidaxomicin) and were randomized 1:1 to placebo or SER-109 groups. ECOSPOR IV was an open-label, single-arm study conducted at 72 US or Canadian sites including 263 participants with rCDI enrolled in 2 cohorts: (1) rollover participants from ECOSPOR III who experienced on-study recurrence diagnosed by toxin EIA (n = 29) and (2) participants with ≥1 CDI recurrence (diagnosed by PCR or toxin EIA), inclusive of the current episode (n = 234). In both studies, the investigational product was administered orally as 4 capsules over 3 consecutive days following symptom resolution after standard-of-care antibiotics. The primary efficacy end point was rCDI (recurrent toxin-positive diarrhea requiring treatment) through week 8. Other end points included CDI recurrence rates and safety through 24 weeks. Results: These 349 participants received at least 1 dose of SER-109 in ECOSPOR III or ECOSPOR IV (mean age 64.2; 68.8% female). Overall, 77 participants (22.1%) enrolled with their first CDI recurrence. Four participants received blinded SER-109 in ECOSPOR III followed by a second dose of open-label SER-109 in ECOSPOR IV. Overall, the proportion of participants who received any dose of SER-109 with rCDI at week 8 was 9.5% (33 of 349; 95% CI, 6.6 %–13.0%), and the CDI recurrence rate remained low through 24 weeks (15.2%, 53 of 349; 95% CI, 11.6%–19.4%), corresponding to sustained clinical response rates of 90.5% (95% CI, 87.0%–93.4%) and 84.8% (95% CI, 80.6%–88.4%), respectively (Fig. 1). Most rollover participants (25 of 29, 86.2%) were from the placebo arm; 13.8% had rCDI by week 8. Conclusions: In this integrated analysis, the rates of rCDI were low and durable in participants who received the investigational microbiome therapeutic SER-109, with sustained clinical response rates of 90.5% and 84.8% at weeks 8 and 24, respectively. These data further support the potential benefit of microbiome repair with SER-109 following antibiotics for rCDI to prevent recurrence in high-risk patients.
Financial support: This study was funded by Seres Therapeutics.
Disclosure: None
Integrated safety analysis of phase 3 studies for investigational microbiome therapeutic, SER-109, in recurrent CDI
- Matthew Sims, Charles Berenson, Stuart Cohen, Elaine Wang, Elizabeth Hohmann, Richard Nathan, Alberto Odio, Paul Cook, Kelly Brady, David Lombardi, Asli Memisoglu, Ananya De, Brooke Hasson, Bret Lashner, Louis Korman, Doria Grimard, Juan Carlos Moises Gutierrez, Barbara McGovern, Lisa Von Moltke
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, pp. s44-s45
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Clostridioides difficile infection (CDI) often recurs in patients aged ≥65 years and those with comorbidities. Clinical trials often exclude patients with history of immunosuppression, malignancy, renal insufficiency, or other comorbidities. In a phase 3 trial (ECOSPOR III), SER-109 was superior to placebo in reducing recurrent CDI (rCDI) risk at week 8 and was well tolerated. We report integrated safety data for SER-109 in a broad patient population through week 24 from phase 3 studies: ECOSPOR III and ECOSPOR IV. Methods: ECOSPOR III was a double-blind, placebo-controlled trial conducted in participants with ≥2 CDI recurrences randomized 1:1 to placebo or SER-109. ECOSPOR IV was an open-label, single-arm study conducted in 263 patients with rCDI enrolled in 2 cohorts: (1) rollover participants from ECOSPOR III with on-study recurrence and (2) participants with ≥1 CDI recurrence, inclusive of the current episode. In both studies, the investigational product was administered as 4 oral capsules over 3 days. Treatment-emergent adverse events (TEAEs) were collected through week 8; serious TEAEs and TEAEs of special interest (ie, bacteremia, abscess, meningitis) were collected through week 24. Results: In total, 349 participants received SER-109 in ECOSPOR III and/or ECOSPOR IV (mean age 64.2; 68.8% female). Chronic diseases included cardiac disease (31.2%), immunocompromised or immunosuppressed (21.2%), diabetes (18.9% ), and renal impairment or failure (13.2%). Overall, 221 (63.3%) of 349 participants who received SER-109 experienced TEAEs through week 24. Most were mild to moderate and gastrointestinal. The most common (>5% of participants) treatment related TEAEs were flatulence, abdominal pain and distension, decreased appetite, constipation, nausea, fatigue, and diarrhea. No participants experienced a treatment-related TEAE leading to study withdrawal. Invasive infections were observed in 28 participants (8%); those with identified pathogens were unrelated to SER-109 species, and all were deemed unrelated to treatment by the investigators. There were 11 deaths (3.2%) and 48 participants (13.8%) with serious TEAEs, none of which were deemed treatment related. There were no clinically important differences in the safety profile across subgroups of sex, race, prior antibiotic regimen, or number of CDI recurrences. No safety signals were observed in participants with renal impairment or failure, diabetes, cardiac disease, or immunocompromised or immunosuppressed individuals. Conclusions: In this integrated analysis of phase 3 trials, SER-109, an investigational microbiome therapeutic, was well tolerated in this vulnerable patient population with prevalent comorbidities. No infections, nor those with identified pathogens, were attributed to SER-109 or product species. This safety profile might be expected because this purified product is composed of spore-forming Firmicutes normally abundant in the healthy microbiome.
Financial support: This study was funded by Seres Therapeutics.
Disclosures: None
Interrelationship between micronutrients and cardiovascular structure and function in type 2 diabetes
- Grace W. M. Walters, Emma Redman, Gaurav S. Gulsin, Joseph Henson, Stavroula Argyridou, Thomas Yates, Melanie J. Davies, Kelly Parke, Gerry P. McCann, Emer M. Brady
-
- Journal:
- Journal of Nutritional Science / Volume 10 / 2021
- Published online by Cambridge University Press:
- 04 October 2021, e88
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
-
Micronutrients are important for normal cardiovascular function. They may play a role in the increased risk of cardiovascular disease observed in people with type 2 diabetes (T2D) and T2D-related heart failure. The aims of this study were to (1) examine micronutrient status in people with T2D v. healthy controls; (2) assess any changes following a nutritionally complete meal replacement plan (MRP) compared with routine care; (3) determine if any changes were associated with changes in cardiovascular structure/function. This was a secondary analysis of data from a prospective, randomised, open-label, blinded end-point trial of people with T2D, with a nested case–control [NCT02590822]. Anthropometrics, cardiac resonance imaging and fasting blood samples (to quantify vitamins B1, B6, B12, D and C; and iron and ferritin) were collected at baseline and 12 weeks following the MRP or routine care. Comparative data in healthy controls were collected at baseline. A total of eighty-three people with T2D and thirty-six healthy controls were compared at baseline; all had micronutrient status within reference ranges. Vitamin B1 was higher (148⋅9 v. 131⋅7; P 0⋅01) and B6 lower (37⋅3 v. 52⋅9; P 0⋅01) in T2D v. controls. All thirty participants randomised to routine care and twenty-four to the MRP completed the study. There was an increase in vitamins B1, B6, D and C following the MRP, which were not associated with changes in cardiovascular structure/function. In conclusion, changes in micronutrient status following the MRP were not independently associated with improvements in cardiovascular structure/function in people with T2D.
Efficacy and Safety of Investigational Microbiome Drug SER-109 for Treatment of Recurrent Clostridioides difficile Infection
- Barbara McGovern, Mathew Sims, Colleen Kraft, Elaine Wang, Kelly Brady, Christopher Ford, O’Brien Edward, Mary-Jane Lombardo, Jennifer Wortman, Kevin Litcofsky, Jennifer Mahoney, Christopher McChalicher, Jonathan Winkler, Sarah Garant, John Aunins, Matthew Henn, Lisa von Moltke
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 1 / Issue S1 / July 2021
- Published online by Cambridge University Press:
- 29 July 2021, p. s5
-
- Article
-
- You have access Access
- Open access
- Export citation
-
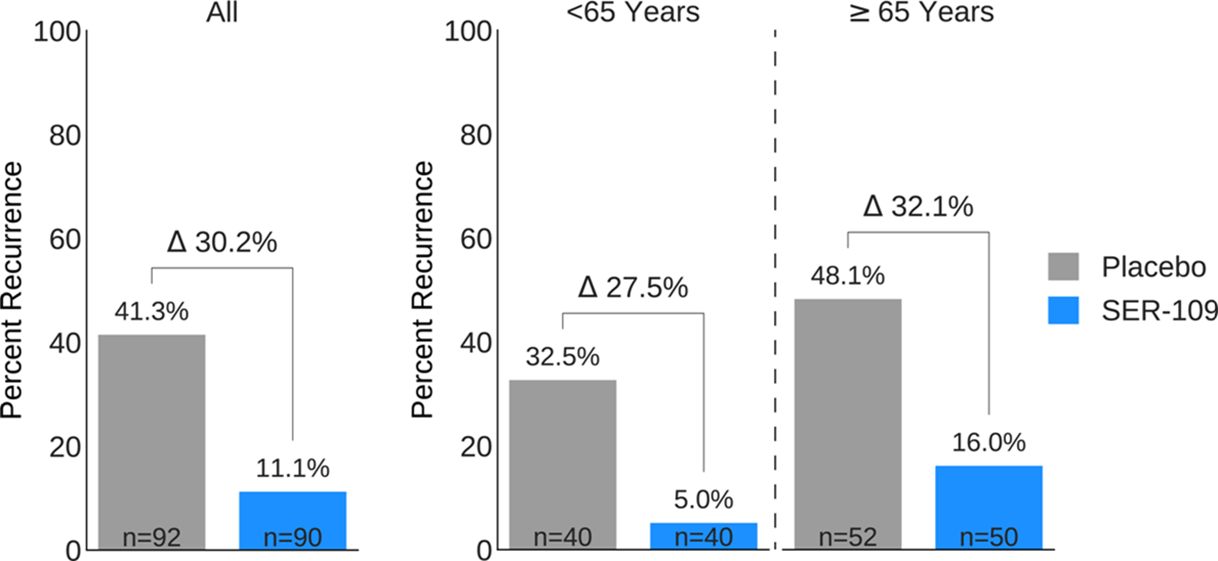
Background: Antibiotics targeted against Clostridioides difficile bacteria are necessary, but insufficient, to achieve a durable clinical response because they have no effect on C. difficile spores that germinate within a disrupted microbiome. ECOSPOR-III evaluated SER-109, an investigational, biologically derived microbiome therapeutic of purified Firmicute spores for treatment of rCDI. Herein, we present the interim analysis in the ITT population at 8 and 12 weeks. Methods: Adults ≥18 years with rCDI (≥3 episodes in 12 months) were screened at 75 US and CAN sites. CDI was defined as ≥3 unformed stools per day for <48 hours with a positive C. difficile assay. After completion of 10–21 days of vancomycin or fidaxomicin, adults with symptom resolution were randomized 1:1 to SER-109 (4 capsules × 3 days) or matching placebo and stratified by age (≥ or <65 years) and antibiotic received. Primary objectives were safety and efficacy at 8 weeks. Primary efficacy endpoint was rCDI (recurrent toxin+ diarrhea requiring treatment); secondary endpoints included efficacy at 12 weeks after dosing. Results: Overall, 287 participants were screened and 182 were randomized (59.9% female; mean age, 65.5 years). The most common reason for screen failure was a negative C. difficile toxin assay. A significantly lower proportion of SER-109 participants had rCDI after dosing compared to placebo at week 8 (11.1% vs 41.3%, respectively; relative risk [RR], 0.27; 95% confidence interval [CI], 0.15–0.51; p-value <0.001). Efficacy rates were significantly higher with SER-109 vs placebo in both stratified age groups (Figure 1). SER-109 was well-tolerated with a safety profile similar to placebo. The most common treatment-emergent adverse events (TEAEs) were gastrointestinal and were mainly mild to moderate. No serious TEAEs, infections, deaths, or drug discontinuations were deemed related to study drug. Conclusions: SER-109, an oral live microbiome therapeutic, achieved high rates of sustained clinical response with a favorable safety profile. By enriching for Firmicute spores, SER-109 achieves high efficacy while mitigating risk of transmitting infectious agents, beyond donor screening alone. SER-109 represents a major paradigm shift in the clinical management of patients with recurrent CDI. Clinicaltrials.gov Identifier NCT03183128. These data were previously presented as a late breaker at American College of Gastroenterology 2020.
Funding: Seres Therapeutics
Disclosures: None
Figure 1.
A Fuller Picture: Evaluating an Art Therapy Programme in a Multi-disciplinary Mental Health Service
- H. Moss, C. Brady, B. Kelly
-
- Journal:
- European Psychiatry / Volume 41 / Issue S1 / April 2017
- Published online by Cambridge University Press:
- 23 March 2020, p. S612
-
- Article
-
- You have access Access
- Export citation
-
Art therapy has a long history in mental health care but requires an enhanced evidence based in order to better identify its precise role in contemporary services. This paper describes an evaluation of an art therapy programme in an acute adult psychiatry admission unit in Ireland. A mixed method research design was used. Quantitative data were collected through a survey of 35 staff members and 11 service-users. Qualitative data included free-text comments collected in the survey and individual feedback from service-users. Both methods aimed to assess the role of art therapy as part of a multidisciplinary mental health service. Thematic content analysis was employed to analyse qualitative data. Staff demonstrated overwhelming support for art therapy as one element within multidisciplinary services available to patients in the acute psychiatry setting, qualitative feedback associated art therapy with improvements in quality of life and individual support, and emphasised its role as a nonverbal intervention, especially useful for those who find talking therapy difficult. Creative self-expression is valued by staff and service-users as part of the recovery process. Recommendations arising from the research include continuing the art therapy service, expanding it to include rehabilitation patients, provision of information and education sessions to staff and further research to identify other potential long-term effects. The low response of staff and small sample in this study, however, must be noted as limitations to these findings.
Disclosure of interestThe authors have not supplied their declaration of competing interest.
Contributors
-
- By Michael H. Allen, Leora Amira, Victoria Arango, David W. Ayer, Helene Bach, Christopher R. Bailey, Ross J. Baldessarini, Kelsey Ball, Alan L. Berman, Marian E. Betz, Emily A. Biggs, R. Warwick Blood, Kathleen T. Brady, David A. Brent, Jeffrey A. Bridge, Gregory K. Brown, Anat Brunstein Klomek, A. Jacqueline Buchanan, Michelle J. Chandley, Tim Coffey, Jessica Coker, Yeates Conwell, Scott J. Crow, Collin L. Davidson, Yogesh Dwivedi, Stacey Espaillat, Jan Fawcett, Steven J. Garlow, Robert D. Gibbons, Catherine R. Glenn, Deborah Goebert, Erica Goldstein, Tina R. Goldstein, Madelyn S. Gould, Kelly L. Green, Alison M. Greene, Philip D. Harvey, Robert M. A. Hirschfeld, Donna Holland Barnes, Andres M. Kanner, Gary J. Kennedy, Stephen H. Koslow, Benoit Labonté, Alison M. Lake, William B. Lawson, Steve Leifman, Adam Lesser, Timothy W. Lineberry, Amanda L. McMillan, Herbert Y. Meltzer, Michael Craig Miller, Michael J. Miller, James A. Naifeh, Katharine J. Nelson, Charles B. Nemeroff, Alexander Neumeister, Matthew K. Nock, Jennifer H. Olson-Madden, Gregory A. Ordway, Michael W. Otto, Ghanshyam N. Pandey, Giampaolo Perna, Jane Pirkis, Kelly Posner, Anne Rohs, Pedro Ruiz, Molly Ryan, Alan F. Schatzberg, S. Charles Schulz, M. Katherine Shear, Morton M. Silverman, April R. Smith, Marcus Sokolowski, Barbara Stanley, Zachary N. Stowe, Sarah A. Struthers, Leonardo Tondo, Gustavo Turecki, Robert J. Ursano, Kimberly Van Orden, Anne C. Ward, Danuta Wasserman, Jerzy Wasserman, Melinda K. Westlund, Tracy K. Witte, Kseniya Yershova, Alexandra Zagoloff, Sidney Zisook
- Edited by Stephen H. Koslow, University of Miami, Pedro Ruiz, University of Miami, Charles B. Nemeroff, University of Miami
-
- Book:
- A Concise Guide to Understanding Suicide
- Published online:
- 05 October 2014
- Print publication:
- 18 September 2014, pp vii-x
-
- Chapter
- Export citation
Distribution of the birds of the Philippines: biogeography and conservation priorities
- A. Townsend Peterson, Lisa G. Ball, Kelly W. Brady
-
- Journal:
- Bird Conservation International / Volume 10 / Issue 2 / June 2000
- Published online by Cambridge University Press:
- 13 November 2000, pp. 149-167
-
- Article
-
- You have access Access
- Export citation
-
The Philippine islands hold a concentration of species diversity and endemism of global importance, yet few studies have analyzed biogeographic patterns or attempted to prioritize areas for conservation within the archipelago. We analyzed distributions of 386 species on 28 Philippine islands and island groups, documenting intense concentration of species richness, especially of endemic species, on the two largest islands, Mindanao and Luzon. Factors identified as influencing species richness included island area, maximum elevation, and Pleistocene patterns of connection and isolation. Reserve systems were developed based on heuristic complementarity algorithms, and compared with the existing Integrated Protected Areas (IPAS) system in the country, showing that IPAS is an impressive first step towards protecting avian diversity in the country. Addition of presently proposed reserves on Palawan and Mindoro would make IPAS a near-optimal reserve design, at least at the level of island representation. Important challenges remain, however, with regard to design of reserve systems within islands to represent complete island avifaunas.





